Despite the landmark passage of the 1938 Federal Food, Drug, and Cosmetic Act...
Exporting Low-Acid Canned Foods to the United States: A Complete Guide to FDA Three-Tier Registration
2026-03-31
Why is three-tiered FDA registration required?
Chinese food manufacturers must complete a three-tiered registration process when exporting low-acid or acidified canned foods to the United States:Food Facility Registration (FFR)、Food bottling plant registration (FCE)andEstablished Process ID (SID)。
According to the FDA's Low Acid Canned Foods Regulation (CFR Title 21 Part 108-113), these foods are subject to stricter regulations than regular foods due to the higher risk of botulism poisoning.
The core reason for three-tier registration
Public Health and Safety
Low-acid foods (pH > 4.6) and acidified foods may grow botulism bacteria if not properly processed in sealed containers, leading to fatal poisoning.
Hot working verification
The FDA requires that every product formulation and processing procedure (scheduling) be scientifically validated to ensure sufficient sterilization effect.
Facility supervision
The bottling facility must comply with CGMP (Current Good Manufacturing Practices) and be subject to regular FDA inspections.
⚠️ Practical Reminder
Many Chinese bottling companies mistakenly believe that they only need to complete the General Food Facility Registration (FFR) to export their products. They only discover that they lack FCE and SID registrations when their goods are detained by U.S. Customs, resulting in serious losses.
What are low-acid and acidified canned foods?
Low-Acid Canned Foods (LACF)
According to CFR Title 21 § 113.3, low-acid canned food is defined as "a sealed container of food in which any component has a pH value greater than 4.6 and a water activity (aw) greater than 0.85".
Common product types:
canned vegetablesCorn, peas, mushrooms, bamboo shoots
Canned meatLuncheon meat, meat sauce, canned chicken
Seafood cannedTuna, salmon, clams
Canned soup: Thick soup, broth
Acidified canned foods
Acidified food refers to "low-acid food products whose final pH value drops to 4.6 or below after the addition of acidic substances (such as vinegar or citric acid)".
Common product types:
Pickled vegetablesPickled cucumbers, pickled peppers, kimchi
Acidified sauceSalsa, ketchup (including vegetable chunks)
Canned fruitSome low-acid fruits undergo acidification treatment.
Why do acidified and low-acid canned foods pose a higher risk of food safety?
Risk of botulism
The most serious threatIt is botulism (Clostridium botulinum poisoning):
Clostridium botulinum spores can multiply and produce deadly toxins in low-oxygen, low-acid (pH>4.6) and normal-temperature environments.
Even trace amounts of botulinum toxin can be fatal.
The sealed environment of canned foods creates ideal anaerobic growth conditions for Clostridium botulinum.
Consequences of insufficient heat treatment
Low-acid foods(pH>4.6) Sufficient high-temperature sterilization is required to kill Clostridium botulinum spores.
Oxidized foodsAlthough adding acid to lower the pH level can inhibit Clostridium botulinum, the acidification process must be uniform and thorough.
Insufficient heat treatment temperature or time, or uneven acidification, can all lead to the survival of pathogenic bacteria.
Historical lessons
Several cases of botulism from canned food occurred in the United States in the 1970s, prompting the FDA to establish a strict regulatory system. Notable cases include:
In 1971, a poisoning incident occurred involving vichyssoise soup from Bon Vivant Soup Company.
Multiple cases of food poisoning from homemade canned food
Explanation of 21 CFR Part 108
Improper handling during the production, processing, or packaging of acidified foods could lead to harmful products entering interstate trade, posing a potential threat to public health. Because the safety risks of such foods are difficult to identify and control in a timely manner once they enter the market, FDA commissioners, based on public health protection considerations, have the authority to require relevant companies to apply for and hold a temporary emergency permit under Section 404 of the Federal Food, Drug, and Cosmetic Act.
The significance of FDA regulation
Register via FFR, FCE, and SID:
Ensure the scientific nature of the manufacturing processThe sterilization process for each product has been validated.
Establish a traceability systemThe source can be quickly located once a problem occurs.
Preventive controlEliminate risks before product launch
Complete explanation of three-tier registration: FFR, FCE, SID
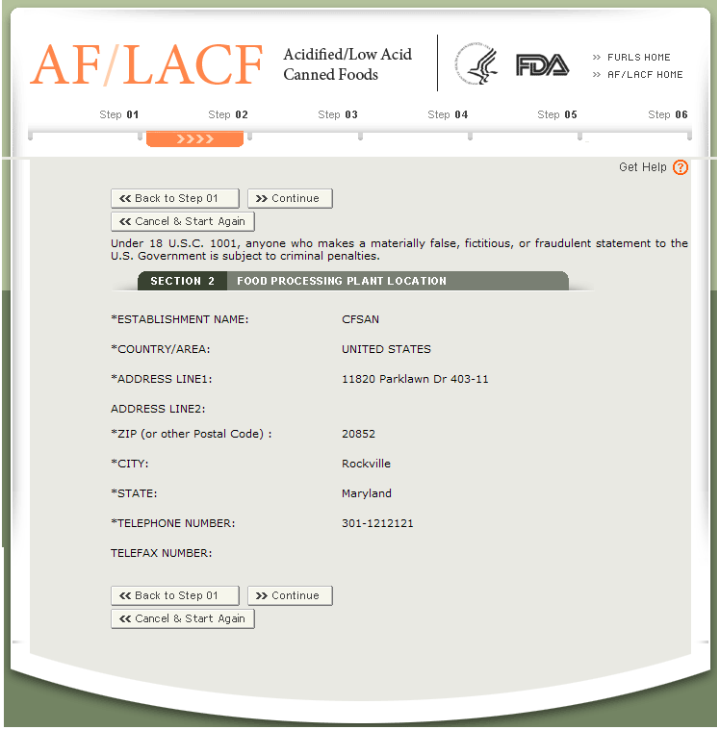
Tier 1: Food Facility Registration (FFR)
Applicable toAll facilities that manufacture, process, package, or store food.
Registration content:
Basic facility information (name, address, contact person)
US Agent Information
Food category (select "Canned food")
Validity periodUpdated every two years (October 1st to December 31st in even-numbered years).
Registration PlatformFDA Food Facility Registration System (FFR)
Second level: Food Canning Establishment (FCE)
Applicable toAll facilities that produce low-acid or acidified canned foods.
Registration content:
Detailed information on bottling processing facilities
List of processing equipment (sterilization autoclave, can sealing machine, etc.)
Quality Control System Description
Processing supervisor qualification certificate (Better Process Control School completion certificate)
Special requirements:
At least one "processing supervisor" with FDA-approved training certification must be assigned.
The facility must meet CGMP requirements.
Registration methodElectronic or paper registration can be submitted or mailed to the FDA.
The third layer: Scheduled Process (SID) registration.
Applicable toFormula for each low-acid or acidified canned product
Registration content:
Product formulation and ingredient ratio
Container type and size
Hot working parameters (temperature, time, pressure)
Processing flow chart
Scientific verification report of the scheduling process (issued by the authoritative processing institution).
Key Concepts"Scheduled processing" refers to heat treatment parameters that have been scientifically calculated and experimentally verified to ensure that products can achieve commercial sterility under specific conditions, that is, to kill all pathogens and spores.
Registration methodElectronic or paper registration can be mailed or faxed to the FDA.
FDA Application Process and Document Preparation for Chinese Bottling Companies
Application order (must be completed in sequence)
Complete FFR food facility registration(Apply online, effective immediately)
Submitting FCE bottling plant registration
Submit SID scheduling and registration
Training requirements for processing supervisors
According to CFR 21 113.10, bottling facilities must assign at least one person who has completed "Better Process Control School" training to supervise heat processing operations.
Training content includes:
Low-acid food microbiology
Principles and calculations of hot working
Equipment Operation and Maintenance
HACCP and CGMP requirements
Practical suggestions
Chinese businesses can send personnel to attend accredited training courses in the United States or Asia, or entrust qualified consultants to serve as processing supervisors.
